With cleanliness knowledge cultivated on semiconductor lines,
We support quality and reliability.
Cultivated on the semiconductor line
In Cleanliness Findings,
We support quality and reliability.
SGV provides comprehensive support for cleanliness at manufacturing sites by leveraging its cleanliness know-how cultivated over half a century of semiconductor manufacturing. We have a solid track record of contributing to yield improvement for over 20 years, especially in the camera module assembly and manufacturing process, which requires a highly clean environment.
In addition to process design and optimization for new packages, yield improvement and defect analysis for existing lines, foreign material control and contamination risk reduction will be thoroughly addressed. In addition, quality control and standardization are strengthened through educational programs and document development. SGV provides Japanese quality support to solve on-site issues and improve long-term reliability.

Cleanliness consulting
Cleanroom Consulting
Based on the know-how cultivated in semiconductor clean rooms, SGV identifies risk factors based on on-site observations and data, and provides integrated support for process design and optimization, condition creation (parameter optimization), defect analysis and prevention of recurrence, and mass production start-up support. We provide integrated support for process design and optimization, condition setting (parameter optimization), failure analysis and recurrence prevention, and mass production start-up support. In addition, we support the establishment of quality control and standardization at the work site through education programs and document maintenance, to ensure that the effects of improvements “continue” with Japanese quality.
Services provided
| Process design/optimization/improvement | New flow design, yield and throughput improvement of existing processes, material and equipment selection support |
|---|---|
| Conditioning (parameter optimization) | Optimization of conditions for key processes, efficient |
| Defect analysis and planning of countermeasures | Root cause identification, assistance in handling complaints, and prevention of recurrence |
| Mass production start-up support | Smooth start-up/transfer of prototypes and evaluations through collaboration between sites |
| Education and Documentation Maintenance | Training, maintenance of technical materials, knowledge sharing |
| Cleaner processes | Minimize contamination risk through process design, equipment improvement, and particle/foreign matter control |
track record
Achievements
Based on our knowledge of process improvement and cleanliness cultivated in the field of semiconductor manufacturing, we provide support aimed at yield improvement and quality stabilization. We will accompany you to a “state of continuous improvement” with on-site data and implementation capabilities.
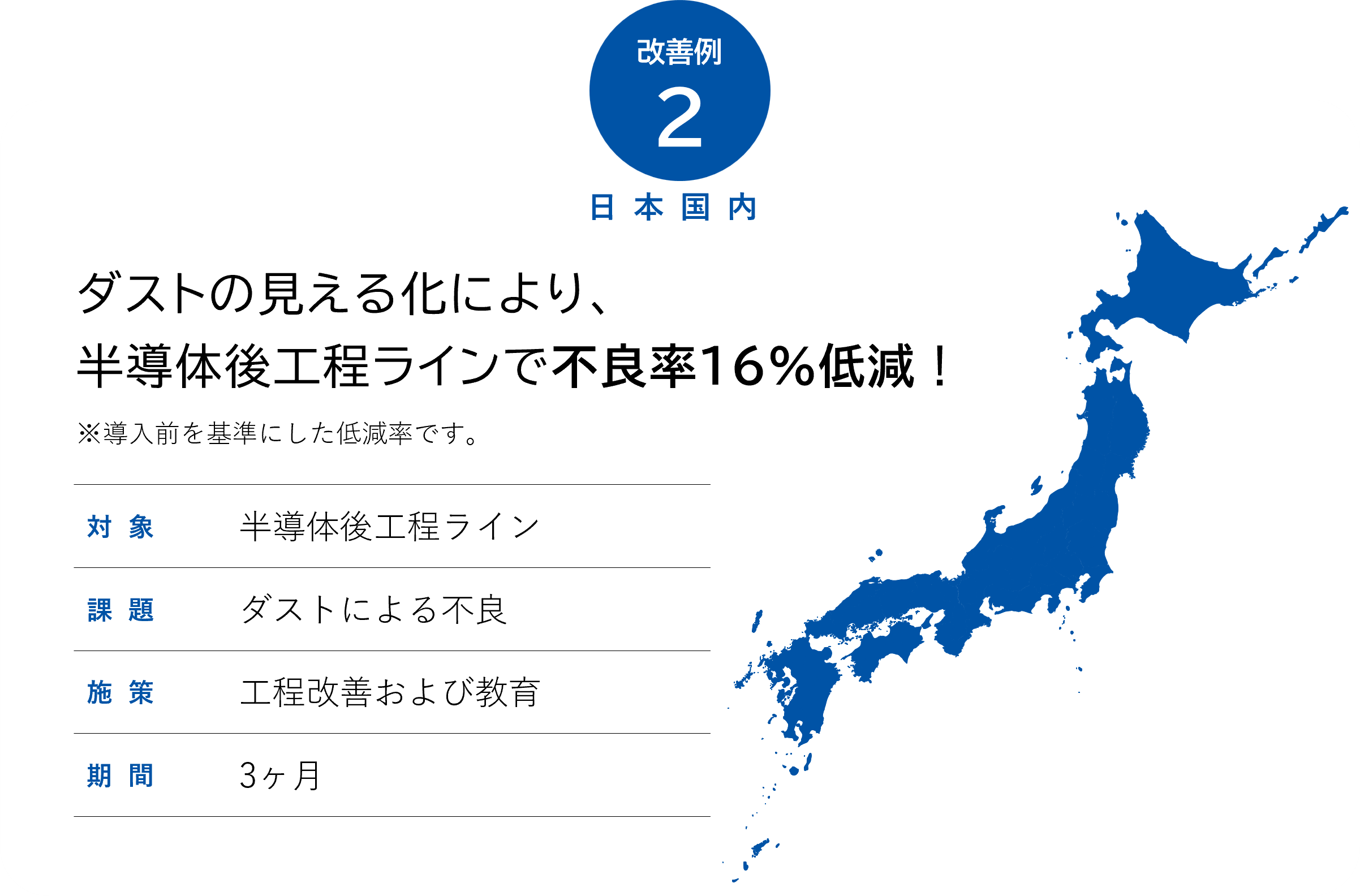
Achievements of yield improvement (typical examples)

Steps to Implement a Clean Process
Steps for Implementing a Cleaning Process
We will not stop with short-term measures, but will also include verification of effectiveness, standardization, and training to create a state in which improvements can continue.

Frequently Asked Questions (FAQ)
Common Inquiries
Yes, it is possible.
SGV applies its knowledge of cleanliness and process improvement cultivated in semiconductor manufacturing to process improvement and quality stabilization in the general manufacturing industry. We can consult with any industry for foreign material control, process variation reduction, yield improvement, etc.
Yes, improvements to existing lines are supported.
Based on on-site observation and data analysis, we propose improvement measures that can be implemented without stopping operation and step-by-step process review. We can also provide partial support for yield improvement and defect reduction.
No problem.
We can handle everything from failure analysis. We identify the root cause of the problem through phenomenon sorting, data analysis, and on-site confirmation, and support the formulation of countermeasures, including prevention of recurrence.
Yes, it is possible.
<br
We will propose realistic cleanliness measures to minimize contamination risk by improving process design, equipment, and foreign material control, without necessarily assuming a clean room.
Yes, we can provide assistance even before the start of mass production.
<br
We support smooth transition to mass production and quality stabilization by providing consistent support from process design, condition creation (parameter optimization), prototyping, and evaluation.



